World appears an aesthetic place due to the colours that are present around us. These are either natural or man-made. Since an ancient times, colours has been playing significant role in human's custom, culture, behaviour, folk religion etc to express their own emotion, feelings and to retain distinct identity as well.
Exploring chemistry of colour ...





In this article, we will discuss,
1. History: From Nature to Synthetics
Natural Dye and Its limitation
Accidental Invention of first Synthetic Dye and Afterward Revolution
2. Basic Terminology to understand Dye Chemistry
Dye vs Pigment
Chromophore and Auxochrome
Substantivity and Reactivity
Bathochromic and Hypsochromic shift
Mordent
3. The Science of Dyes: How Colours Work
4. Different Types of Synthetic Dyes
Azo-dyes
Disperse-dyes
Direct-dyes
Reactive-dyes
Vats-dyes
Sulfur-dyes
Acid-dyes
Basic-dyes
5. Toxicological concerns and Need of Greener, Smarter, Safer Dyes
6. Application of Dyes
7. Case Studies: Metanil Yellow Adulteration in Turmeric and Detection Methods
8. Properties of Synthetic Dyes
9. Improving qualities of Natural Dyes
10. Conclusion : Why It Matters
1. History: From Nature to Synthetics
Natural Dye and its limitation :
Humans have been obsessed with colour for millennia. Before discovery of synthetic dyes, they used to extract natural dyes from different biotic organism such as indigo leaves for deep blue, madder roots for red, or cochineal insects for a vivid scarlet, through grinding, boiling, or fermenting, then applied to fabrics or leather. But this process was time-consuming and less-durable as it can be faded with time if not taken care properly. Some of the natural dyes are fugitive and need a mordant for the enhancement of their fastness properties. Some of the metallic mordants are hazardous.
Accidental Invention of first Synthetic Dye and Afterward Revolution:
Then, a big leap came in 1856 after 28 years of first synthesis of organic compound ( Urea by Friedrich Wöhler), when William Henry Parkin, 18-year old British chemist, was trying to synthesise quinine (a anti-malarial drug) but ended up with making first synthetic dyes of brilliant purple, named mauveine, from coal tar.
William Henry Perkin – Pioneer in Synthetic Organic Dyes (1856)
This incident sparks a revolution. By the late 19th century, synthetic dyes—brighter, cheaper, and more reliable—overtook natural ones, fuelling the textile boom of the Industrial Age. Today, dye chemistry is a powerhouse, versatile from fashion to medicine, chemical industry of synthetic dyestuffs to the pharmaceutical industry, which improved the quality of life for the general population.
Ancient civilizations in India, Egypt, and China begin using natural dyes from plants (e.g., indigo), animals (e.g., cochineal), and minerals, extracting colors by boiling or fermenting for fabrics and art.
German chemist Friedrich Wöhler synthesizes urea, marking the start of organic chemistry. This paves the way for future dye research by showing that organic compounds can be made in labs, inspiring synthetic dye development.
British chemist William Henry Perkin accidentally creates mauveine, the first synthetic dye, from coal tar while trying to make a malaria drug.
2. Basic terminology to understand Dye Chemistry
Before it is very to know basic definition of dyes and how it is different from pigment.
a. Dyes vs. Pigments
Dyes are soluble molecules that penetrate and bond with materials, like fabric or paper. Pigments, however, are insoluble particles that sit on surfaces, coating rather than soaking in.
For instance, take Holi (the Indian festival of colors) where its gulal—colours powders used in Holi's celebration—are pigments, temporarily dusting skin and clothes.
Dyes, by contrast, might tint the fabrics worn during Holi, offering a lasting stain. This difference shapes their use and chemistry.
b. Chromophore and Auxochrome
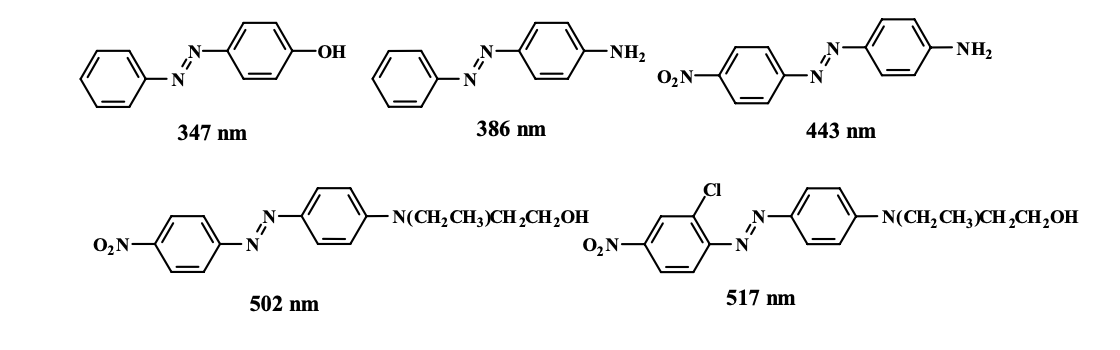
Chromophore can be described as the part of a dye molecule responsible for emitting colour by absorbing specific light wavelengths in range of 420 nm - 750 nm such as (-N=N-) linkage in azo-dyes.
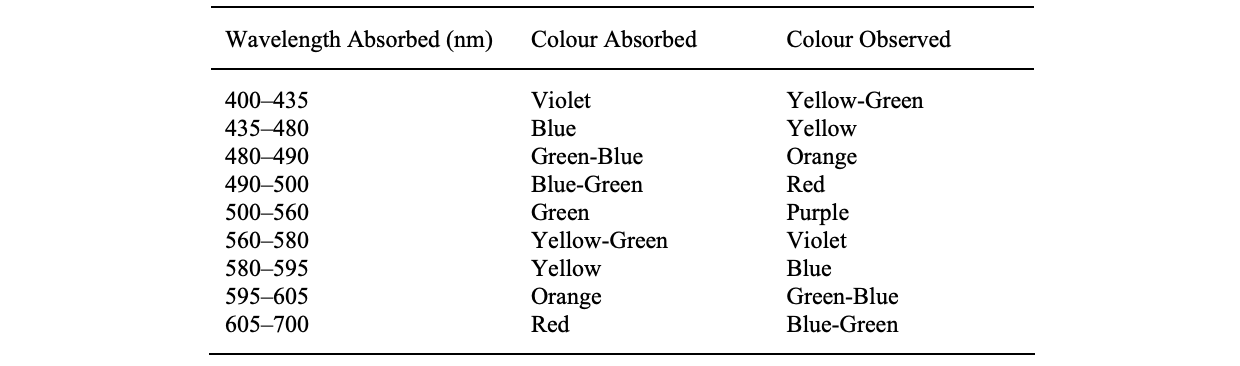
While, Auxochrome is a group that enhances the color or changes the color of the chromophore such as carboxylic acid,
sulfonic acid, amino, and hydroxyl groups. While these are not responsible for colour but their presence can shift the colour of a colourant and they are most often used to influence
dye solubility.
c. Substantivity and Reactivity
Substantivity can be defined as the ability of a dye to be retained by a fibre or material naturally while reactivity is the ability of a dye to chemically bond with a fibre or material.
Hence, dyes must be designed that have
- Greater affinity for the substrate than the
medium (usually water) from which it is applied - Substantivity
- A high degree of permanence (e.g. stability to fading upon exposures to water (wet fast) and/or sunlight (light fast) - Reactivity
d. Bathochromic Shift and Hypsochromic Shift
In bathochromic, there is a shift in the absorption spectrum of a molecule to longer wavelengths (lower energy) due to the introduction of conjugation or other structural changes that stabilize the excited state. This results in a red shift in the observed color (deeper color : orange to red)
While hypsochromic shift is totally opposite of bathochromic shift, where a peak shifts towards shorter wavelengths (higher energy) due to the disruption of conjugation.This results in a blue shift in the observed color (lighter color : blue to green)
- Adding groups of increasing electron-donating ability to the azobenzene structure has a bathochromic effect, making a deeper color
- Increasing the number of electron-withdrawing groups conjugated with the electron-donor has a bathochromic effect.
- Increasing the length of the conjugated system in compound can causes a significant bathochromic shift.
As ability of electron-releasing group increase or number of electron-withdrawing in conjugation with electron-releasing group increase, leading to shift toward longer wavelength and so making it deeper color.
e. Mordent
Natural dyes are substantive, requiring a mordant to fix to the fabric, and prevent the colour from either fading with exposure to light or washing out. These compounds bind the natural dyes to the fabric. They are of three types :
- Metallic mordants: Metal salts of aluminium, chromium, iron,
copper and tin are used.
- Tannins: Myrobalan and Sumach are the commonly used tannins
employed as mordants in the dyeing of textile fibres.
- Oil mordants: Oil mordants are used mainly in the dyeing of turkey red colour from maddar. The main function of the oil mordant is to form a complex with alum used as the main mordant. The sulfonated oils, which possess better metal binding capacity than the natural oils due to the presence of sulfonic acid group, bind to metal ions forming a complex with the dye to give superior fastness and hue.
3. The Science of Dyes: How Color Works
Organic compound will posses color only if
- absorb light in the visible spectrum (400–700 nm)
- have at least one chromophore (colour-bearing group)
- have a conjugated system, i.e. a structure with alternating double and single bonds.
- exhibit resonance of electrons, which is a stabilising force in organic compounds.
When any one of these features is lacking from the molecular structure, the colour is lost.
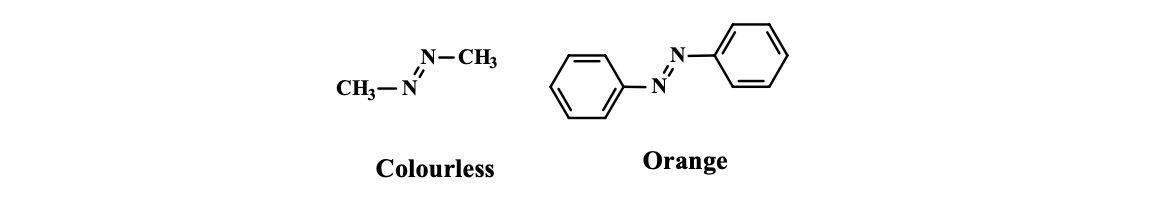
Placement of an azo-group between methyl groups produces a colourless compound, while a yellow-orange colour is obtained when the azo-group is placed between aromatic rings due to conjugation.
4. Different Types of Synthetic Dyes :
a. Azo Dye
Known for vivid yellows, oranges, and reds, these dyes contain the -N=N- group in chromophore, making them versatile for textiles, food, and even tech applications like solar cells.
I have already discussed method of preparation for dyes in previous article, you can check through below given link
(Link of article : Two application of Nitrous Acid in Organic Chemistry)
b. Disperse Dye
Used for dyeing hydrophobic fibers like polyester, nylon, acetate and triacetate fibers etc and are poorly soluble in water. Hence, disperse dyes have no affinity for hydrophilic polymers such as cellulose, which makes them unsuitable for colouring cotton, cellophane, and paper but quite suitable for poly(ethylene terephthalate) and cellulose acetate.
c. Direct Dye
Cellulosic materials like cotton, rayon, linen, cellophane, and paper are hydrophilic, needing water-soluble dyes that resist washing out (e.g., during laundering or coffee spills).
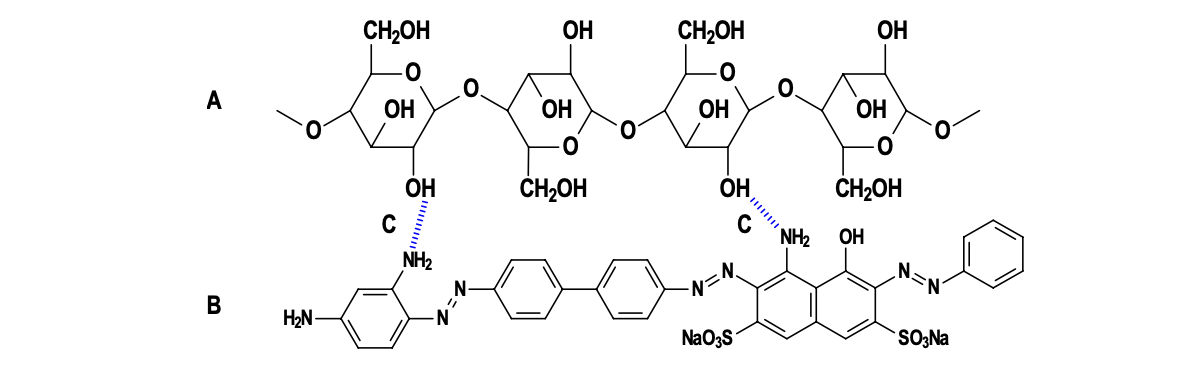
Direct dyes are water-soluble, linear molecules (e.g., benzidine-based) align with cellulose via hydrogen bonding and can dye cellulose fibers without the need for mordants (substances that help the dye bind to the fiber)
They’re easy to apply but often lack wet fastness, fading when washed.
Representation of H-bonding (C) between a cellulose (A) and a direct dye (B)
d. Reactive Dye
These dyes form covalent bonds with the fibers, resulting in high color fastness and are commonly used for dyeing cotton and other cellulose fiber. Hence, they show better comparative durability against light and washing.
e. Vats Dye
Naturally insoluble (e.g., indigo), they’re made water-soluble via a “vatting” reduction process, penetrate the fiber, then oxidize back to insoluble form. This locks in color, though indigo’s small size allows fading, as seen in denim.
f. Sulphur Dye
Polymeric and sulfur-containing, these dyes follow a similar reduction-oxidation process to vat dyes. Their structures are less defined but offer affordable, wash-resistant earthy tones for cotton.
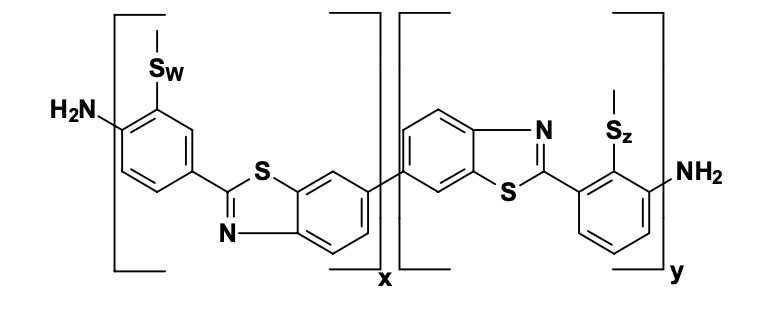
g. Acid Dye
A dyes bearing a negative charge are used for polyamides carrying positive charge such as nylon and proteins such as wool, silk, and leather – forming ionic bonds within the polymer matrix.
Schematic representation of dye–polymer binding via ionic bonding on nylon
These dyes have little to no affinity for polyester, cellulosic, or cationic polymers, since such substrates cannot form an ionic bond with them.
h. Basic Dye
Basic dye will work same as acid dye does except nature of charge carrying by substrate or dye and polymer for which dye is used.
A dyes bearing a positive charge are used for poly(acrylonitrile) carry a negative charge – forming ionic bonds within the polymer matrix.These dyes have no affinity for polyester, cellulosic, or polyamide polymers, since such substrates cannot form an ionic bond with them.
Previously, we have discussed Mauveine (the first synthetic dye) which is a basic dye used for dyeing silk. This takes advantage of the presence of carboxylate (–CO2 - ) groups in silk and wool.

5. Toxicological concerns and Need of Greener, Smarter, Safer Dyes
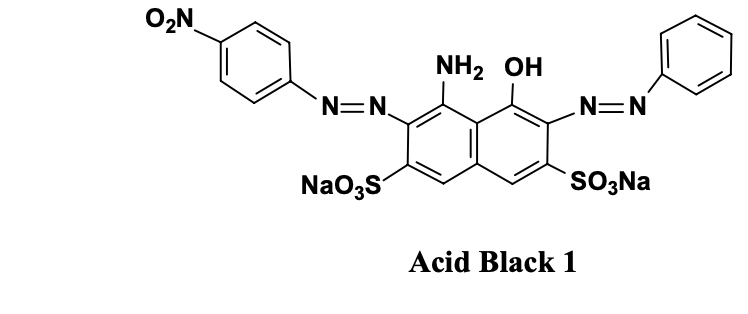
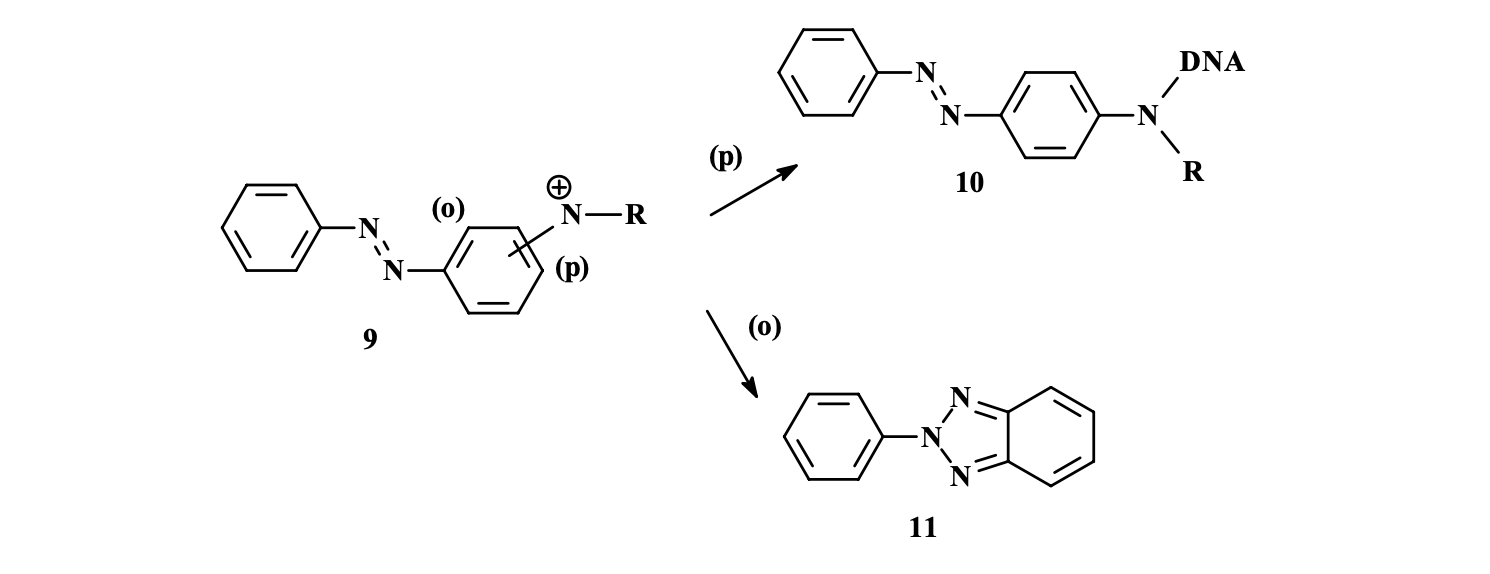
Synthetic dyes must pose minimal health risks, avoiding raw materials like carcinogenic aromatic amines (e.g., benzidine), which are linked to bladder cancer via DNA-damaging metabolites.
Examples of carcinogenic aromatic amines and azo-compounds
Even though using non-carcinogen dye, metabolism can yield genotoxic amines because safe dyes may break down into harmful byproducts. Studies show hydrophobic azo dyes (e.g., with methyl groups) increase carcinogenicity, especially para-isomers that form DNA-reactive nitrenium ions, unlike safer ortho-isomers that cyclize harmlessly.
Reaction pathways for nitrenium-ion formed from metabolism of azobenzene
(a) Through pathway (p), how DNA (protein cell) is corrupted or mutated due to nitrenium ion
(b) cyclisation by ortho-isomers through pathway (o)
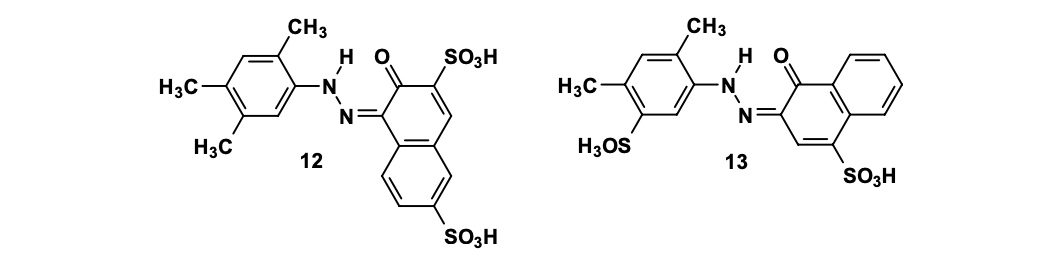
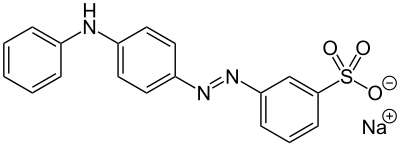
Now, coming to Hydrophilic azo dyes that how affect metabolism ? Well, it vary too !! those yielding lipophilic amines (e.g., 2,4,5-trimethylaniline) are riskier than those producing water-soluble sulfonated amines.
Carcinogenic (left) and non-carcinogenic (right) water-soluble monoazo-dyes
Some of the chemicals found in synthetic dyes are mercury, lead, chromium,copper, sodium chloride, toluene, and benzene. Exposure to large doses of these substances can be toxic and can have severe effects in the human body. Also, most dyes are not biodegradable, meaning they can persist in the environment for a long time, leading to long-term environmental impacts.
Hence, toxicological concerns can shape this evolution and there is a need to priorities sustainability, innovation, and safety in dye chemistry. Bioengineered bacteria produce natural dyes like indigo, cutting resource use, while synthetic dyes become biodegradable, reducing pollution. Smart dyes shift with heat or light, and nanoscale dyes refine precision. Modern design thus balances affinity, eco-friendliness, and toxicology, phasing out toxic metals and prioritizing skin-safe, non-mutagenic options.
6. Application of Synthetic Dye
Textile
Synthetic dyes are commonly used in the textile industry to color fabrics such as cotton, silk, wool, and synthetic fibres like polyester, nylon, and acrylic.

Printing
They are also used in the printing industry to produce a range of printed materials such as magazines, newspapers, books, and packaging.

Cosmetics
Synthetic dyes are used in the cosmetics industry to add color to products such as lipsticks, eye shadows, and nail polish.

Food
In the food industry, synthetic dyes are used to add color to products such as candies, baked goods, and beverages.

Plastic
These dyes are used in the production of plastics to add color and improve the aesthetic appeal of products such as toys, containers, and consumer electronics.

Pharmaceuticals
In the pharmaceutical industry, synthetic dye is used to add color to medications for ease of identification and differentiation.
7. Case Studies: Metanil Yellow Adulteration in Turmeric and Detection Methods
Metanil Yellow, a synthetic azo dye, has been used to adulterate turmeric powder to mimic its natural yellow for economic gain. Two case studies illustrate simple detection methods and their impact:
- Case Study 1: Sodium Hydroxide Spot Test in India
A practical test uses a few drops of sodium hydroxide (NaOH) solution on turmeric powder. Pure turmeric turns reddish-brown, while adulteration with Metanil Yellow produces a violet or purple hue due to the dye’s reaction with the base. In a local market survey, 85% of 20 samples showed this violet color, indicating widespread adulteration linked to mild toxicity risks like digestive irritation. This method is easy for consumers but may miss low dye levels, suggesting follow-up with experts.
On adding few drops of NaOH to Turmeric stem (left) and standard metanil yellow (right) giving reddish yellow and violet solution respectively
- Case Study 2: Chemical Testing with Hydrochloric Acid in India
A household test adds hydrochloric acid to a turmeric sample, turning the solution pink if Metanil Yellow is present. A survey of 22 market samples found 91% adulterated, associated with nausea and stomach disorders. This low-cost approach raises awareness but lacks precision for small concentrations, highlighting the need for advanced verification.
Appearance of pink color in turmeric indicates the presence of metanil yellow adulteration.
Note: These methods are adapted from standard food safety protocols, such as those in the FSSAI’s “Detect Adulteration with Rapid Test” guidelines (2017), and reflect common practices in India for detecting synthetic dyes in spices.
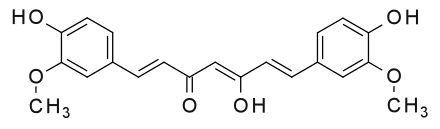
Chemical Structure of Metanil Yellow Dye (left) and Curcumin present in turmeric (right), both exhibiting yellow color
8. Properties of Synthetic Dyes
The properties of synthetic dye depend on the specific type of dye, but in general, synthetic dyes have the following properties:
- Brightness and consistency: Synthetic dyes are known for their bright and vibrant colors, which are consistent across different batches.
- Stability: These dyes are stable and do not fade easily in light or with exposure to other environmental factors.
- Affordability: They are generally more affordable than natural dyes, making them a cost-effective option for large-scale production.
- Chemical properties: Different types of synthetic dyes have different chemical properties that make them suitable for different types of materials and applications. For example, acid dyes are used for coloring protein fibres like wool and silk, while disperse dyes are used for coloring synthetic fibers like polyester.
- Resistance to water and other chemicals: These dyes are generally resistant to water and other chemicals, making them suitable for use in a wide range of applications.
9. Improving quality of Natural Dyes
Natural dyes, like those from indigo, sometimes fade when exposed to sunlight because their color-giving part (the chromophore) gets damaged by light—a process called photooxidation. To fix this, scientists have binded up the dye with transition metals (like iron or copper) to form a protective complex, helping the color last longer in the sun. They’ve also made natural dyes better at staying on fabric after washing by using eco-friendly helpers called mordants, such as alum, stannic chloride, stannous chloride, and ferrous sulphate. Adding tannins (natural compounds from plants) with these mordants makes the dye stick even more. When metal salts mix with tannins, they change how tannins absorb light and make them water-insoluble, so the fabric holds the color through washes. These simple method bring natural dyes closer to the durability of synthetic ones!
Conclusion : Why It Matters
Dye chemistry is transforms our daily lives, from the vibrant clothes we wear to the safe foods we eat and important for filling consciousness, while driving scientific breakthroughs. For everyone, it’s the magic behind the colors that brighten festivals like Holi or protect us from harmful adulterants. For chemists, it’s a fascinating puzzle of molecules and reactions, pushing innovation toward greener, safer solutions. This blend of art and science continues to paint our world with endless possibilities.
Emphasising on how dyes bring joy and consciousness to daily life and expressing emotion







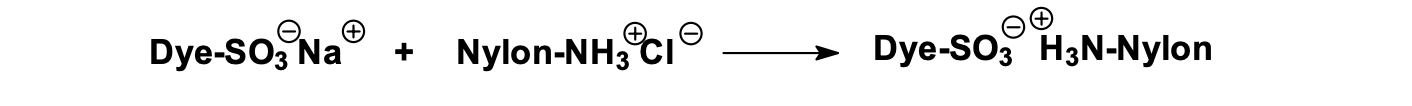




Exploring the World of Dyes : The Chemistry of Color